|
(Radioactive tritium is neither a fissile nor a fissionable material.) There are no enriched materials in a fusion reactor like ITER that could be exploited to make nuclear weapons. Limited risk of proliferation: Fusion doesn't employ fissile materials like uranium and plutonium. The activation of components in a fusion reactor is anticipated to be low enough for the materials to be recycled or reused within 100 years, depending on the materials used in the "first-wall" facing the plasma. No long-lived radioactive waste: Nuclear fusion reactors produce no high activity, long-lived nuclear waste. Its major by-product is helium: an inert, non-toxic gas. No CO₂: Fusion doesn't emit harmful substances like carbon dioxide or other greenhouse gases into the atmosphere. (Terrestrial reserves of lithium would permit the operation of fusion power plants for more than 1,000 years, while sea-based reserves of lithium, used in a fusion reactor in its Li-6 isotope form, would fulfil needs for millions of years.) A critical challenge is how to breed and recover tritium reliably in a fusion device. Deuterium can be distilled from all forms of water, while tritium will be produced during the fusion reaction as fusion neutrons interact with lithium. Millions of years: Fusion in ITER will require two elements: deuterium and tritium. Fusion has the potential to provide the kind of baseload energy needed to provide electricity to our cities and our industries. The following advantages make fusion worth pursuing.Ībundant energy: Fusing atoms together in a controlled way releases nearly four million times more energy than a chemical reaction such as the burning of coal, oil or gas and four times as much as nuclear fission reactions (at equal mass). The fossil fuels that shaped 19th and 20th century civilization can only be relied on at the cost of greenhouse gases and pollution.Ī new large-scale, sustainable and carbon-free form of energy is urgently needed. © EUROfusion)The next decades are crucially important to putting the world on a path of reduced greenhouse gas emissions.īy the end of the century, demand for energy will have tripled under the combined pressure of population growth, increased urbanization and expanding access to electricity in developing countries. (An artist's impression of the European fusion power plant design. a number of advantages make fusion worth pursuing. The values above are the total energy yield, not the energy delivered to a consumer.Sustainability, abundant fuels, no long-lived waste. This figure is dated and probably high, but it gives a basis for comparison. Then they are expressed in terms of a nominal per capita U.S. Both the single event energy and the energy per kilogram of fuel are compared. Nuclear binding energy curveĭeuterium-tritium fusion and uranium-235 fission are compared in terms of energy yield. 
Its average binding energy per nucleon is exceeded only by 58Fe and 62Ni, the nickel isotope being the most tightly bound of the nuclides. Iron-56 is abundant in stellar processes, and with a binding energy per nucleon of 8.8 MeV, it is the third most tightly bound of the nuclides. The iron limitThe buildup of heavier elements in the nuclear fusion processes in stars is limited to elements below iron, since the fusion of iron would subtract energy rather than provide it. Whereas an atomic transition might emit a photon in the range of a few electron volts, perhaps in the visible light region, nuclear transitions can emit gamma-rays with quantum energies in the MeV range. The binding energies of nucleons are in the range of millions of electron volts compared to tens of eV for atomic electrons. 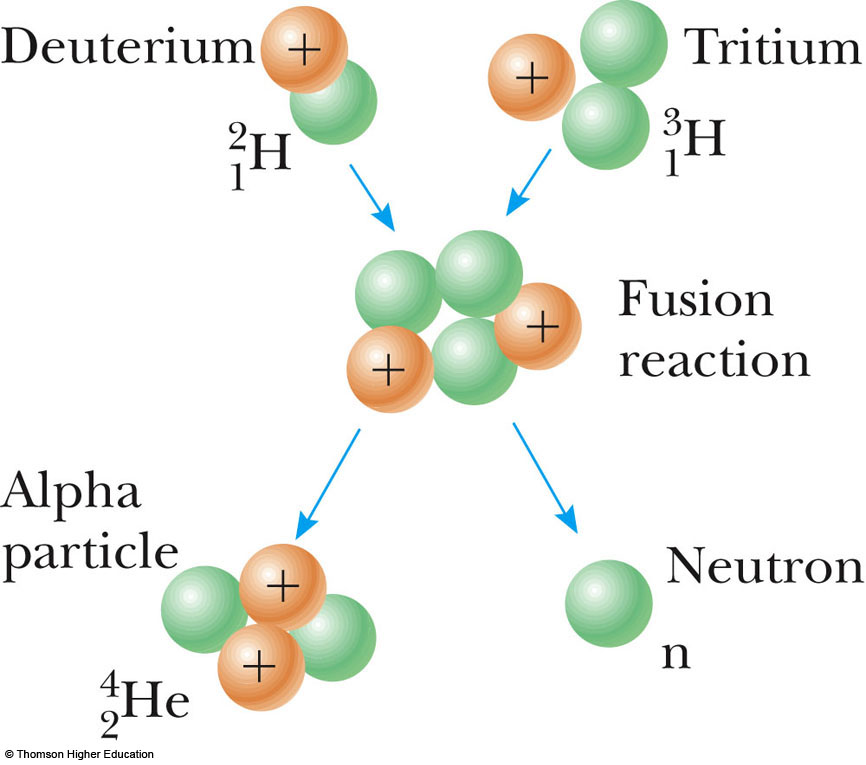
The fact that there is a peak in the binding energy curve in the region of stability near iron means that either the breakup of heavier nuclei (fission) or the combining of lighter nuclei (fusion) will yield nuclei which are more tightly bound (less mass per nucleon). 
The binding energy curve is obtained by dividing the total nuclear binding energy by the number of nucleons. The nuclear binding energies are on the order of a million times greater than the electron binding energies of atoms. The comparison of the alpha particle binding energy with the binding energy of the electron in a hydrogen atom is shown below. The enormity of the nuclear binding energy can perhaps be better appreciated by comparing it to the binding energy of an electron in an atom. This binding energy can be calculated from the Einstein relationship: Nuclear binding energy = Δmc 2įor the alpha particle Δm= 0.0304 u which gives a binding energy of 28.3 MeV. The difference is a measure of the nuclear binding energy which holds the nucleus together. Nuclei are made up of protons and neutrons, but the mass of a nucleus is always less than the sum of the individual masses of the protons and neutrons which constitute it. Nuclear Binding Energy Nuclear Binding Energy
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
